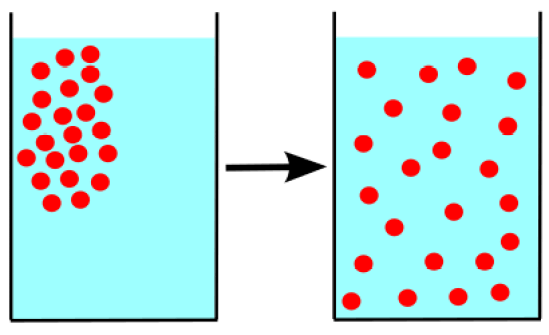
Osmosis and diffusion can equalize the concentration of molecules, and both are spontaneous processes that don’t need any external energy. Osmosis and diffusion move from higher concentration to lower concentration, but both have some differences, which we can discuss today to understand the different effects of these two other processes. These two processes are more common in our daily lives. And play a vital role in moving in or out of the cell.
What is osmosis?
Osmosis is the movement of a solvent, e.g., water molecules, from the lower to the highest to equalize the concentration. In this process, the solvent passes through a semipermeable membrane to balance the attention. This process doesn’t want any energy.
Osmosis is very common in plant cells. The plant body is a semipermeable membrane that allows the water to move in. Still, it doesn’t allow some ions and carbohydrates to move in. the water moves into the plant to equalize the concentration of solute and solvent.
What is diffusion?
Diffusion is the movement of particles (gas, solid, liquid) from a higher concentration to a lower concentration to balance in the medium. No cell wall or barrier is present in this process, so the molecule can move quickly to balance in a container. Simple diffusion doesn’t take energy, while passive mediated diffusion needs energy.
When we use perfume, its fragrance starts spreading in the whole room, a diffusion process.
Differences Between Osmosis And Diffusion

| Osmosis | Diffusion |
| The water or other liquid solute from higher concentration to the lower concentration to equalize the concentration. | The particles may be gas, water, or liquid pass from higher concentration to lower to balance in the medium. |
| This needs liquid-like water to pass. | It doesn’t need liquid to pass. It can move freely. |
| Osmosis requires semipermiable membrane to move. | It doesn’t require any semipermeable membrane to move. |
| Osmosis depends on solute potential. | Diffusion doesn’t depend on solute potential, pressure, and liquid. |
| The flow of particles occurs in one direction. | The flow of particles occurs in all directions. |
| The solute molecules cannot become equal on both sides. | The diffusion particles spread in the medium and equalize the concentration. |
| Occur only in similar types of solutions. | Occur in all types of particles. |
| The process can be stopped by applying external pressure or force. | This can not be stopped. |
| However, It involves only solvents to move one side to the other. | So, It involves all types of particles to move one side to the other. |
| So, Hydrostatic pressure and turgor pressure oppose osmosis. | Therefore, Hydrostatic and turgor pressure can not apply to diffusion. |
Similarities of Osmosis And Diffusion
In both the osmosis and diffusion processes, the molecules move from higher to lower to balance the concentration.
Both are passive transport because they don’t need external energy.
They Both can equalize the concentration.
Both are natural processes.
Also read: how many calories in a cup of tea