Before discussing the measurement of ions using an ion electrode, I will explain a little about the ions in an aqueous solution.
In “Basics of pH measurement-the concept of acidity and alkalinity “, “Water is expressed by the molecular formula H 2 O, and most of them exist in the form of a very stable molecule H 2 O, but only a small part. hydrogen ion H Tasu and hydroxide ion OH – was said to exist in the separate form “.
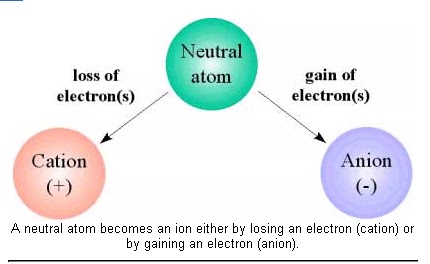
What is an ion?
Due to the presence of water in this way at room temperature, water dissolves many substances. (Although there are some substances that do not dissolve, such as the saying of “water and oil” …). Let’s take a look at the difference in properties between the case where sugar is dissolved in water and the case where salt is dissolved.
When sugar, especially high-purity granulated sugar, is dissolved in water, the ease of flow of electricity (correctly, electric current) in this sugar water is not much different from that of the original water. On the other hand, when salt is dissolved, electricity becomes very easy to flow in this saltwater. Why.
Ionic Bond
The reason for sugars in water (e.g., glucose C 6 H 12 O 6 ) does not have the ion, salt (NaCl) completely ions, i.e. sodium ions Na + and chloride ion Cl – since it Because there is. In other words, the presence of these ions made it easier for the current to flow. The detailed reason omitte, but the sodium atom Na originally electronic E – sodium one let go by the ion Na+ The likely, whereas chlorine atom Cl electron e – 1 or takes in chloride ion Cl a – has a property of easily become. Salt (NaCl) combination of Na + and Cl – that electrically attracted to each other. This form of the substance icalled an “ionic bond”.
On the other hand, saccharides compounds formed by sharing a part of each electron (valence electron) in a slightly complicated form of the carbon atom, hydrogen atom and oxygen atom, that “shared compound”, and even if they dissolve in water, they in the water. It exists without divided into ions. Apparently, sugars and salts are also soluble in water, but there is a big difference depending on whether they become ions in water.
Bottom Line
Human blood, contained in such lymph, sodium ions Na play a physiological effect. On the important role such as osmotic +, potassium ions K +, chloride ion. Cl -, bicarbonate ion HCO 3 -, etc., medicine, physiology It called “electrolyte”. In the technical term of the field (the term electrolyte is a little broader in the field of physics. And chemistry, and in short, it understood as the concept of all various ions).
Ions with a positive charge called “cations” and ions with negative charges called “anions”. The number of electrons that enter and leave when becoming an ion called the “valence”. And counted as monovalent, divalent, or trivalent. Na + is a monovalent cation, Cl – is a monovalent anion, Ca 2+ is a divalent cation. SO 4 2- is a divalent anion, Fe 3+ is a trivalent cation, PO 4 3- is a trivalent anion. There are also ions with higher valences. The sum of the charges of the cations and anions present. In the water is always equal, so they are electrically neutral when viewed from the outside.
We hope that you will get an overview of “what is ion” from the above explanation.
As will describe in the next section, the ion electrode developed. For the purpose of easily measuring these ions in the form of ions.
Also read: how much are lip fillers